Michael Halassa is professor of Psychiatry and Biomedical Engineering at Virginia Tech. Halassa is also a board-certified and practicing psychiatrist who specializes in treatment of psychotic disorders. His clinical research is focused on identifying novel precision targets based on emerging pharmacology and neurostimulation. He has been consistently funded by the U.S. National Institutes of Health and has received several fellowships and prizes throughout his career—most notably, the Vilcek Prize for Promise in the Biomedical Sciences (2017), an award given to immigrant scientists who have made extraordinary contributions to American society. This essay was originally published on his Substack.
I spend a good portion of my time thinking about what schizophrenia is. The diagnosis is built on a foundation of psychotic experiences: voices no one else hears, beliefs held with certainty despite disconfirming evidence, thinking that loses its coherent structure. Psychiatry has defined this constellation in many ways that can feel made up, but this is a common human attribute; just consider money, government or most concepts we agree on as a civilization. There is urgency with psychiatric categories, however; the past two decades have given us perturbations powerful enough to expose the limits of our inherited categories.
A 22-year-old man with no psychiatric history begins using high-grade cannabis daily. Within months, he becomes convinced his neighbors are monitoring him. He hears voices commenting on his actions. His family brings him to the emergency room, where he is diagnosed with cannabis-induced psychotic disorder. He stops using cannabis, receives antipsychotic medication, and improves. But six months later, off all substances, he relapses with the same paranoid beliefs and auditory hallucinations. The diagnosis is revised to schizophrenia. Did the cannabis cause the schizophrenia, unmask latent vulnerability, or was this always going to happen regardless?
A 28-year-old woman experiments with a high-potency stimulant at a party. She does not sleep for three days. By day four, she is certain her thoughts are being broadcast and that strangers on the street are coordinating against her. She is hospitalized, treated, and recovers fully. She never uses stimulants again and remains well for years. Was this drug-induced psychosis, or a brief psychotic episode that the stimulant triggered? Would she have developed schizophrenia eventually, or was this an isolated event determined entirely by pharmacological perturbation?
A 37-year-old woman with no history of mental illness presents to the inpatient unit floridly psychotic. She is fixated on the idea that she has a rare, contagious disease and poses a danger to other people. The fixation is disorganized, difficult to follow, without clear narrative structure. It is not immediately obvious what precipitated the episode. Speaking with her parents reveals she has spent months isolated in her room, talking to ChatGPT for hours each day. When asked directly about her ChatGPT use, she leans forward and asks, “Is my ChatGPT talking to you?” After starting antipsychotic medication, her psychosis resolves over two weeks. As she improves, she gains insight: “I see now how this might have triggered my altered thinking.” Was this a psychotic disorder precipitated by extended Large Language Model (LLM) interaction, or would she have decompensated regardless?
By Diagnostic and Statistical Manual (DSM) criteria, all three of these patients could eventually carry a diagnosis of schizophrenia. Yet the experience of psychosis itself is heterogeneous enough across these cases that it is unclear we are talking about the same phenomenon. Do these represent the same disorder reached through different precipitants, or different disorders that converge on a shared diagnostic label? The answer determines whether precision psychiatry for psychosis is possible and whether our inherited diagnostic categories are adequate.
What We Inherited
When Emil Kraepelin distinguished dementia praecox from manic-depressive illness in the late 19th century, he was likely working with a more constrained set of cases than we see today.
Kraepelin’s distinction was based on clinical course. Dementia praecox began early and deteriorated progressively. Manic-depressive illness followed an episodic pattern with better outcomes. The key assumption was that course reflected underlying disease process: one trajectory, one disease. For the patients he observed in late 19th century psychiatric hospitals, often severely ill and institutionalized, this framework worked reasonably well.
Eugen Bleuler renamed dementia praecox as schizophrenia and shifted focus from course to symptoms. He emphasized loosening of associations, ambivalence, and the “split” in normal mental integration. He grouped together patients with hallucinations, delusions, disorganized thinking, and negative symptoms under a single diagnostic entity. The patients Bleuler saw shared enough phenomenological overlap that treating them as variations on a common pathology made sense. He retained Kraepelin’s core assumption: this was a unitary disease process arising from some biological defect.
Kurt Schneider later proposed first rank symptoms to sharpen the diagnosis further: specific types of auditory hallucinations (voices commenting on one’s actions, voices conversing), experiences of thought insertion or withdrawal, delusions of control. These were meant to separate schizophrenia from other conditions that gave rise to psychotic experience. Again, this reflected clinical experience with a patient population that may have been more constrained than what we observe today.
The DSM codified this inheritance into a checklist: two or more symptoms from a specified list, present for a specified duration, causing functional impairment. Hallucinations, delusions, disorganized speech, disorganized behavior, and negative symptoms were treated as interchangeable indicators of the same underlying condition.
This progression from Kraepelin through Bleuler and Schneider to the DSM all presumed the same thing: schizophrenia is one disease. Patients who meet criteria have the same underlying pathology, even if their specific symptom profiles differ.
But the world they observed no longer exists. In Kraepelin and Bleuler’s era, psychosis arose primarily from genetic and developmental vulnerabilities, perhaps precipitated by infection, trauma, or severe stress. Today we have pharmacological and environmental factors powerful enough to induce psychotic states that were unavailable a century ago. The routes to psychosis have multiplied, and with them, the heterogeneity of the phenomenon we are trying to capture with a single diagnostic category.
The Rise of Powerful Environmental Perturbations
High-grade cannabis is not the cannabis of previous generations. Modern strains are bred for THC concentrations exceeding 20%, delivering sustained, potent activation of cannabinoid receptors throughout the brain. Population studies show clear dose-response relationships between cannabis potency and psychosis risk, particularly in adolescence.
Modern stimulants produce dopaminergic surges that far exceed anything available in Kraepelin’s era. These compounds can induce florid paranoid psychosis in individuals with no prior psychiatric history and no obvious genetic loading. The psychosis often resolves when the drug clears, but in some cases it persists, raising questions about whether the perturbation unmasked latent vulnerability or directly altered circuit function in a self-sustaining way.
And now we have large language models. The 37-year-old woman who asked if her ChatGPT was talking to me had spent months in isolation, hours each day in dialogue with the system. When she recovered, she herself described it as something that “triggered” her altered thinking. The role of the LLM was not immediately obvious from her presentation. It had to be discovered through careful history-taking with her family. What made this case striking was not just that an LLM interaction preceded psychosis, but that she developed a genuine belief that it was a coherent agent with access to information beyond what appeared on her screen.
These cases represent routes to psychosis that were rare or absent in the early 20th century. Cannabis existed, but not at these concentrations and not with this level of targeted receptor activation. Stimulants existed, but not compounds engineered for extreme potency. Interactive generative models did not exist at all. I suspect the heterogeneity we now observe in psychosis clinics reflects, at least in part, new ways for brains to fail that were not available to previous generations.
A State Space for Psychosis
To understand why heterogeneity matters, we need to think about state spaces and how different levels of description relate to each other.
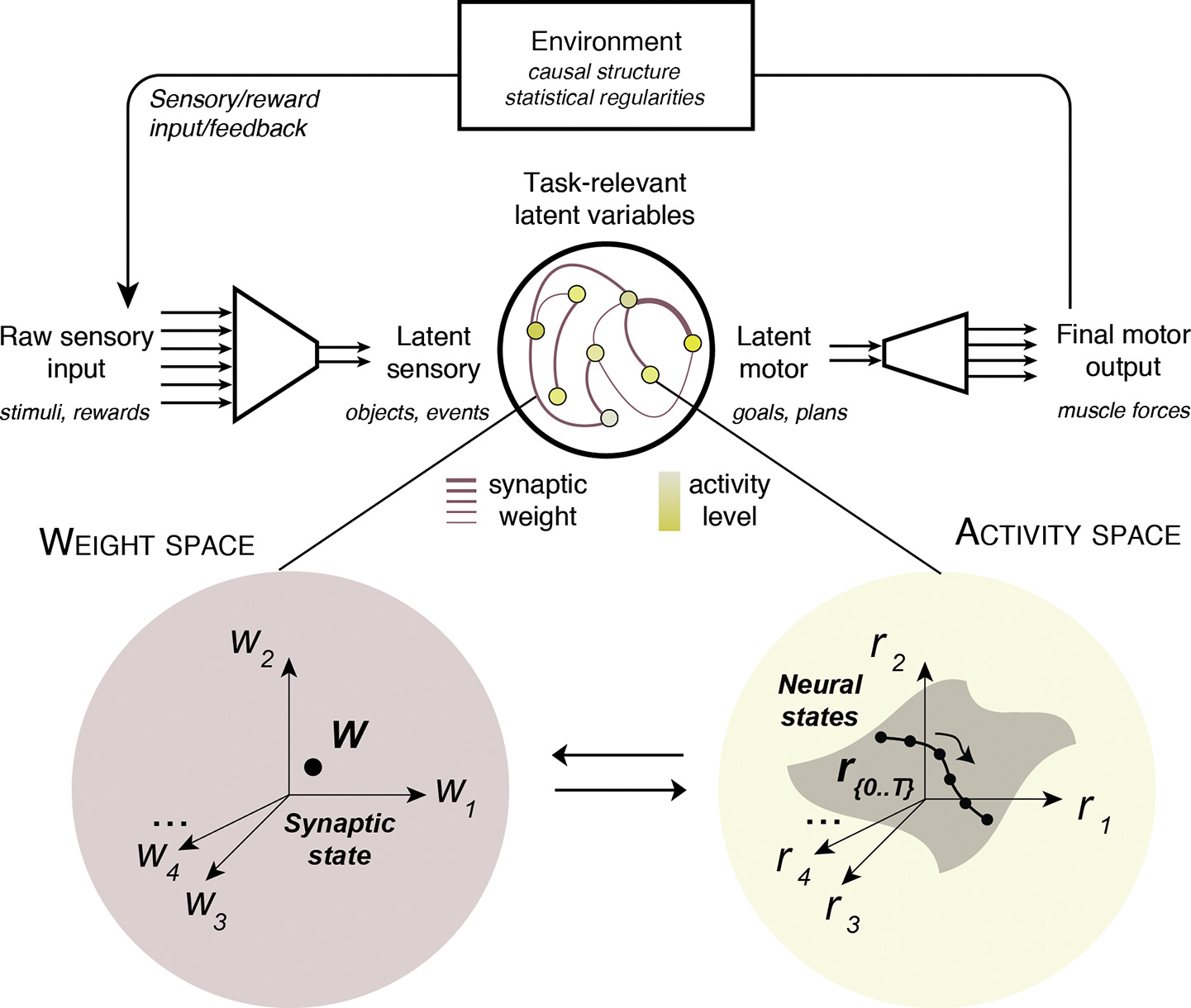
Start with neural state space: the collection of all possible patterns of neural activity. Each point represents a unique pattern of activity indexed by the neurons involved (Please see illustrations and references at the end of this section). Now consider experiential state space: the collection of all possible subjective experiences. Psychotic experiences occupy particular regions of this experiential space. We can assume there exists a mapping between these spaces, such that specific neural states manifest as specific psychotic experiences.
The neural state dynamics can be specified by the parameters of the underlying system: connection strengths between neurons, the magnitude of incoming signals, how much the system amplifies those signals, and the patterns of activity it receives over time, among others. These parameters determine which regions of the neural state space are occupied, how they evolve and, consequently, which experiences are generated. Here is a key insight: different parameter settings can produce similar trajectories in the neural and experiential spaces. Depending on the circuit, doubling synaptic connection strengths might produce roughly the same experiential output as doubling input magnitude or increasing gain control. Therefore, psychosis may arise through genetic alterations leading to aberrant synaptic weights, gain control or plasticity. Drugs may alter these same parameters. LLMs might have an impact similar to gain control via additive inputs that would be difficult to generate outside of this particular feedback loop.
The explosion of underlying parametric perturbations may have also rendered the clinical features of psychosis more heterogeneous. In other words, new perturbations may be pushing the system into previously unvisited or rarely visited regions of the neural and experiential state spaces. Such perturbations may not have existed during Kraepelin’s time, and if true, his understanding of schizophrenia would truly be different than what we are faced with now.
If we are grouping together patients who occupy genuinely different regions of the experiential state space, reached through different perturbations of underlying neural parameters, then the category of schizophrenia captures even less commonality than we thought. An important question we might want to consider is: are we observing the same neural and experiential state space regions Kraepelin did, or are there now new regions that are popping up thanks to the rise of powerful environmental perturbations?
Figure 1 from Sohn et al., 2020 Trends Neurosci. A Network Perspective on Sensorimotor Learning. This illustrates the idea of state spaces, shown here as synaptic weights (left) and neural activity patterns (right). You can imagine a similar relationship between neural states (parameterized by the particular type of measurement(s)) and experiential states (parametrized as computational subtypes similar to the ones in the next section).
Toward Computational Subtypes
If schizophrenia groups together patients who arrived at psychosis through different routes, an important question is whether we can identify meaningful subtypes based on how neural representational systems are altered. Algorithmic circuit psychiatry offers a framework for thinking about this, moving beyond the trial and error approach we currently rely on with medications.
Consider three hypothetical computational subtypes:
Precision weighting alterations. Some patients may struggle to calibrate how much confidence to place in their internal predictions versus external sensory evidence. When internal predictions are weighted too heavily, internally generated signals can be mistaken for external perceptions, producing hallucinations. People have developed behavioral paradigms to measure this tendency and relate it to auditory hallucinations through a Bayesian framework. If this framework is correct, interventions targeting the underlying process might have therapeutic utility (via neurostimulation, medications or otherwise).
Belief updating rigidity. Other patients may show intact precision weighting but impaired belief updating. They might register prediction errors, but fail to have them update their internal models appropriately. This could produce delusional beliefs that persist despite clear disconfirmation. Computational modeling of choice behavior and learning tasks might characterize how flexibly patients update their beliefs. If this characterization proves meaningful, interventions could focus on metacognitive training that explicitly teaches patients to recognize when evidence contradicts their beliefs.
Hierarchical inference breakdown. Still other patients may have difficulty maintaining the hierarchical structure of representations. Higher-level beliefs about context and meaning should constrain lower-level perceptual inferences. When this hierarchy cannot be maintained (perhaps due to capacity limitations), the system might default to lower-level strategies and lack the ability to generalize. Tasks measuring context effects on perception and source monitoring could potentially identify these failures. Interventions might target restoration of hierarchical structure through cognitive remediation focused on context integration.
These categories are not mutually exclusive, and individual patients likely show mixed profiles. But they illustrate how computational characterization could cut across traditional phenomenology. Two patients with paranoid delusions might have fundamentally different computational profiles: one with aberrant precision weighting, another with rigid belief updating. Current nosology groups them together. Computational phenotyping distinguishes them.
How the three cases from the beginning (and others) map onto these computational profiles (or a linear combination thereof) remains unknown. That is precisely the work that needs to be done: developing tools to measure these processes reliably enough to characterize individual patients.
The path forward requires developing these measurements into clinically deployable tools. This is not a distant future. Research groups are already using behavioral tasks, computational modeling, and neuroimaging to characterize how individual patients process information, update beliefs, and represent uncertainty. The question is when these tools become reliable and practical enough to guide treatment decisions.
The Path Forward
Schizophrenia remains useful as a pragmatic category. It allows clinicians to communicate about patients, access treatment algorithms, and organize care. Antipsychotic medications help many people with the diagnosis, regardless of which route brought them to psychosis. Eliminating the category before we have valid alternatives would leave patients worse off.
What schizophrenia becomes depends on whether we can develop precision measurements that characterize how individual brains are computationally altered. The hypothetical subtypes outlined above suggest the direction: measure belief updating flexibility, precision weighting, hierarchical inference, and other algorithmic processes through behavioral tasks and computational modeling. Use these measurements to identify the neural systems involved and guide interventions tailored to specific alterations rather than relying on trial and error.
High-grade cannabis, stimulants, and LLMs create routes to psychosis that were rare or absent when Kraepelin and Bleuler built their nosology. The heterogeneity we observe today may have crossed their threshold for what they would have called a single entity. Building measurement tools precise enough to distinguish these failures and target them with corrective interventions is the work underway. Until computational phenotyping becomes as practical as the DSM checklist, we should keep using schizophrenia while building toward something better. The question is not if, but when schizophrenia will be no more and what types of entities will replace it.
Disclaimer: The clinical vignettes are informed by real patient encounters but have been substantially modified, combined, and fully de-identified to protect privacy. No described scenario corresponds directly to any specific individual I have treated.